 They exhibit symmetries, such as fivefold symmetry, that are forbidden in ordinary crystals. Quasicrystalline solids display novel symmetries in which the atoms are arranged in quasiperiodic fashion-i.e., in patterns that do not repeat at regular intervals. They include glasses, plastics, and gels. 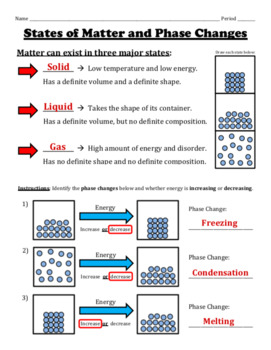
Noncrystalline solids are those in which atoms and molecules are not organized in a definite lattice pattern. Practically all metals and many other minerals, such as common table salt (sodium chloride), belong to this class. Crystalline solids have a very high degree of order in a periodic atomic arrangement. Solids are generally divided into three broad classes- crystalline, noncrystalline ( amorphous), and quasicrystalline. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!. 
Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
